Due to their fixed location, plants must adapt to environmental cues by changes in growth and development - in essence changing their physical form - to ensure survival. The developmental plasticity of plants is unparalleled by any other life form. The primary factor influencing plant responses to environmental cues is the intricate interplay of plant hormones (phytohormones). Like hormones in animals, phytohormones act in cells and tissues distant from their site of origin, involving three essential steps for function: synthesis, transport, and reception.
While synthesis and reception of phytohormones have been the focus of extensive research, knowledge regarding transport remains limited and fragmented. Unlike the case for humans, phytohormones must rely on specialized transporters to cross cell membranes for both export from the producing cell and import into the target cell where reception takes place.
Plant-PATH focuses on two antagonistic pairs of phytohormones, whose distribution orchestrates essential phenotypic traits: auxins and cytokinins that balance root or shoot growth, and abscisic acid (ABA) and gibberellic acid (GA) that balance germination and dormancy of seeds
Plant-PATH wishes to disentangle the complexity of phytohormone transport, and detail the molecular mechanisms dictating local phytohormone concentrations in a time- and location-dependent manner. The center aims to unlock a new perspective on hormone transport, resulting in tools to rewire hormone transport in plants to create new functionalities.
In the long-term, this work is hoped to foster development of strategies for sustainable and climate-resilient agriculture. Thereby, instead of optimizing crops for specific stresses like prolonged drought or heat, as has been attempted for centuries, Plant-PATH will lay the scientific foundation and pioneer research directions towards a future, where crops with a dynamic hormonal response can be engineered. For instance, this involves activating one hormone transporter while inhibiting another, allowing crops to adapt rapidly to the fluctuating temperature and water availability that is characteristic of climate change.
The Pedersen's lab work take part in elucidating the molecular mechanisms that control the movement of key plant hormones and sugars, processes that are central to plant growth, development, and environmental adaptation. Their work focuses on membrane transport proteins that govern the distribution of phytohormones—particularly auxin and cytokinin—as well as the allocation of sugars between plant tissues.
Auxin and cytokinin are two fundamental phytohormones that shape nearly every aspect of plant life. Auxin directs processes such as organ formation, vascular patterning, and responses to light and gravity, while cytokinin regulates cell division, shoot development, senescence, and stress responses. The precise spatial and temporal distribution of these hormones depends on specialized transport proteins embedded in cellular membranes. The Pedersen lab investigates how these transporters recognize their substrates, how they undergo conformational changes during transport, and how their activity is regulated within different cellular contexts.
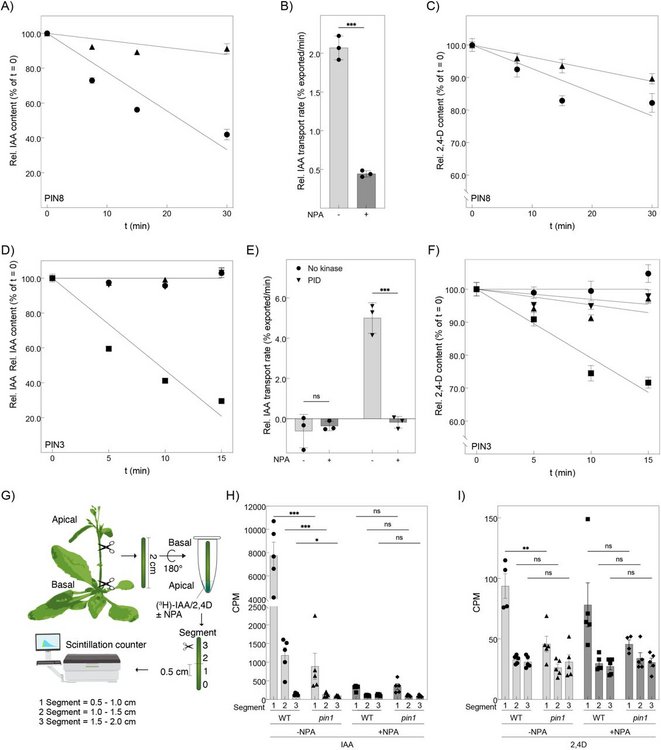
A major focus of this work is the PIN-FORMED family of auxin transporters. Some PIN proteins are regulated at the plasma membrane through phosphorylation of a long, intrinsically disordered loop that acts as a molecular gate for auxin export. Other PIN variants lack this regulatory region and function constitutively inside the cell, particularly at the endoplasmic reticulum, where they control intracellular auxin distribution. By generating targeted mutations and analyzing their effects on transport activity, the lab dissects how structural features determine transporter regulation and function.
In addition to hormone transport, the Pedersen lab studies sugar transport, which is essential for distributing energy from photosynthetic source tissues to non-photosynthetic sink tissues such as seeds, fruits, and pollen. They have characterized sucrose transporters (SUC/SUTs), which mediate long-distance carbon allocation, as well as Sugar Transport Proteins (STPs), which import glucose into cells that lack direct symplastic connections. These transport systems also play important roles in stress responses, including drought tolerance and defense against pathogens.
To address these questions, the lab combines structural biology approaches—primarily cryo-electron microscopy, crystallography, and molecular dynamics simulations—with functional transport assays in yeast, Xenopus oocytes, and solid-supported membrane electrophysiology systems. This integrated strategy connects molecular structure to biophysical function, providing fundamental insights into how transporters operate in living plants.
Overall, the Pedersen lab seeks to define the molecular principles that govern hormone and sugar distribution in plants. This knowledge advances our understanding of plant development and stress adaptation and provides a mechanistic foundation that can ultimately support efforts to improve crop performance and resilience.
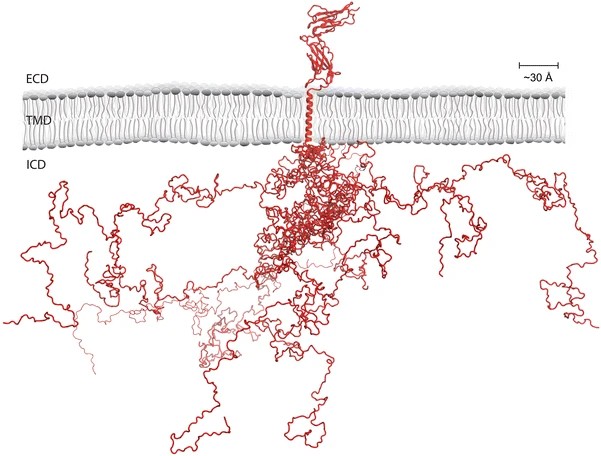
Research in Magnus Kjærgaard group focus on understanding how intrinsically disordered regions (IDRs) of proteins work. IDRs defy the classical protein-structure function paradigm by being functional without folding into a fixed three-dimensional structure. Instead function emerges from a highly heterogeneous ensemble of conformations defined by the protein sequence. Ultimately they hope to be able to engineer new functions into proteins via their IDRs for applications in biotech.
Many hormone transporters have large intrinsically disordered regions that regulate their transport activity. This is most clearly exemplified by the long-loop PINs that have 300 residue long intra-cellular loop that regulates transport activity and couples to other proteins. They believe that by understanding the molecular details of these regulatory mechanisms we will be allowed to re-wire the regulation of plant hormone transport. In Plant-PATH, Magnus’ group investigates how intrinsically disordered regions of hormone transporters regulate transport activity using biophysical techniques including integrative structural biology and de novo protein design.
Experimental techniques in the research group span from theoretical modelling to molecular cell biology, but are mostly focused on biochemistry and molecular biophysics. Their group also administrate the “Biophysics and Biochemistry core facility” at the Department of Molecular Biology and Genetics and are heavy users of its instruments.
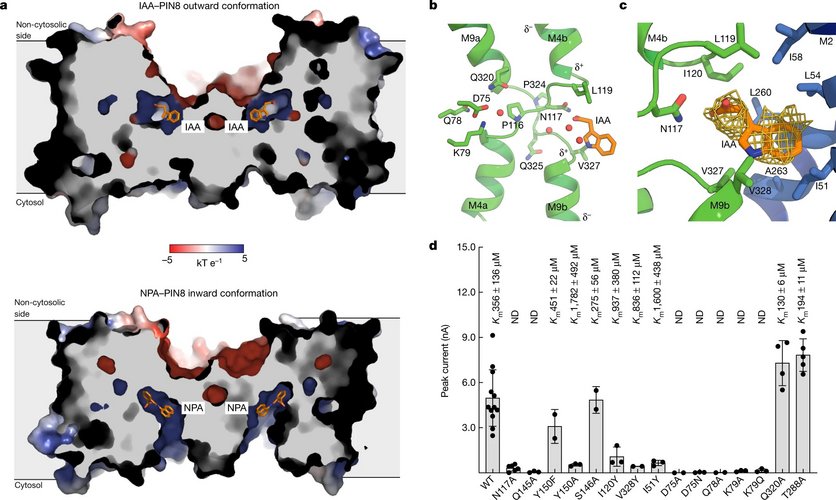
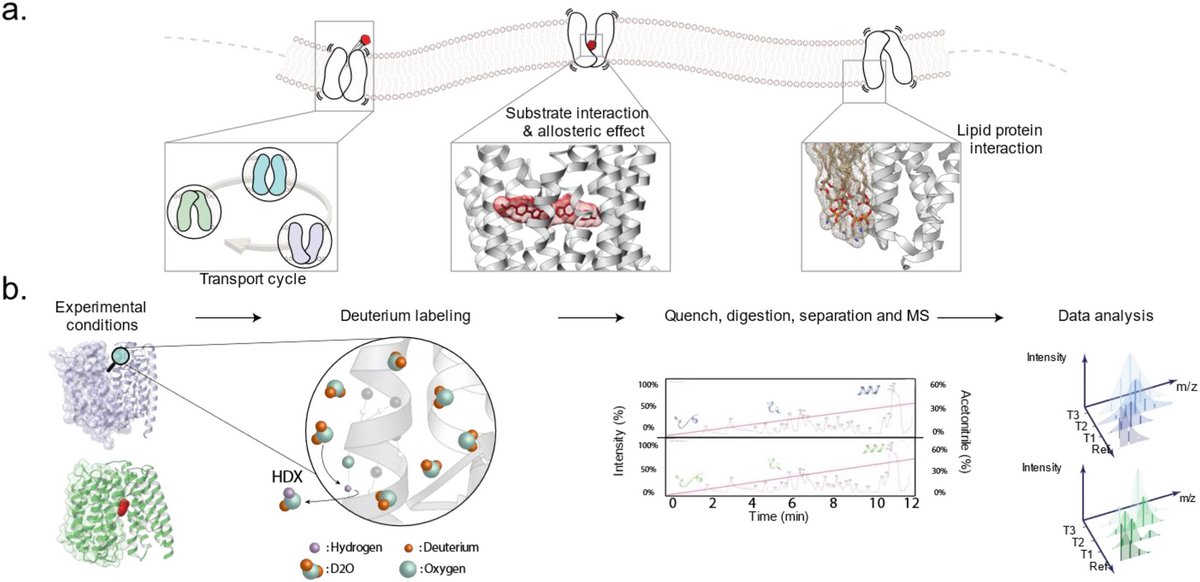
Research in Chloé’s group focuses on understanding how membrane transporters work at the molecular level, and on finding ways to modulate their activity for therapeutic benefit. Transporters are proteins embedded in cellular membranes that move nutrients, metabolites, and drugs across in and out of the cell. They play a crucial role in physiology, but also in disease, as they often determine whether drugs reach their targets. Despite their importance, transporters remain poorly understood because they are dynamic, challenging to purify, and difficult to study with classical structural biology approaches.
In the laboratory at Université Libre de Bruxelles, Chloé’s group combines advanced biophysical and biochemical methods to uncover the dynamic principles that govern transporter function. They have developed a strong expertise in hydrogen–deuterium exchange mass spectrometry (HDX-MS), which allows them to map conformational changes of transporters in solution. They integrate this with single-molecule FRET, cryo-electron microscopy, double electron–electron resonance spectroscopy, and electrophysiological assays such as solid supported membrane (SSM) recordings. By combining these complementary methods, they can capture transporter motions across multiple timescales and directly link them to transport activity. A central theme of their research is to understand how transporters are regulated by their lipid environment.
The group has investigated bacterial multidrug efflux pumps, which underlies antibiotic resistance, human solute carriers which are key drug transporters, and now plant hormone transporters that shape plant phenotypes and responses to stress. Their goal is not only to reveal fundamental mechanisms, but also to identify novel sites for pharmacological or biotechnological modulation.
Ultimately, their research aims to establish transport dynamics as a new dimension in drug discovery and plant biotechnology. By targeting conformational equilibria rather than static structures, they hope to design innovative adjunct therapies that improve drug delivery, overcome resistance, and expand treatment options in neurological diseases, while also informing strategies for crop improvement and resilience.
The research in Ulrich Hammes group focusses on the functional characterization of transport proteins on the biochemical and biophysical level and in the plant system. They investigate transporters for assimilates, mainly sugars and amino acids as well as phytohormones. The main phytohormones are auxin, cytokinin, brassinosteroids, gibberellic acid and abscisic acid. They are also interested in the regulation of these transporters on the posttranslational level and investigate how kinases activate transporters.
To investigate the biochemical and biophysical properties they use heterologous expression systems like yeast or Xenopus laevis oocytes to perform flux studies using labelled substrates. Ulrich’s group host one of the largest Xenopus facilities in Germany and theses oocytes are also used to characterize electrogenic transporters by two electrode voltage clamps. They also use solid supported membrane-based electrophysiology to characterize reconstituted recombinant protein in liposomes.
To characterize the transporters in plants they use confocal microscopy and other microscopy approaches to demonstrate the expression and subcellular localization of GFP fusion proteins or native protein by immunocytochemistry. They also use complementation of plant mutant phenotypes and bioassays to demonstrate the physiological significance of the finding established in the biochemical/biophysical approaches.
Through this work Ulrich’s group aim to understand the interplay of import and export of assimilates and especially hormones into and out of plant cells and over long distances to answer the question how transport processes contribute to the remarkable developmental plasticity of plants.