Mapping of genetic properties affecting the degree of liver injury
An international research team led by Professor Jacob George of Storr Liver Centre in Sydney, Australia - with the participation of researchers from Aarhus University Hospital and Aarhus University - has mapped the human genetic properties affecting the degree of liver injury in patients with hepatitis C virus infection.
Patients infected with Hepatitis C virus (HCV) have a chronic infection (inflammation) in the liver that causes scar tissue formation (fibrosis), which may lead to the development of cirrhosis of the liver. The disease can be treated with antiviral medicine, but the patients are not always cured as they respond differently to the medical treatment.
Previous results from an international research team led by Rune Hartmann from Aarhus University have shown that the outcome of treatment depends, inter alia, on the individual's genetic heritage. In the new article, the Australian research team studies nearly 2000 liver biopsies from HCV infected patients. The results from these studies show that the same genes also determine the rate of hepatic inflammation and hence liver injury (measured as fibrosis) in HCV patients.
Specifically, the researchers found that there are variations in our genes for the protein Interferon Lambda 4 (IFNL4), which determines the extent of the patients’ response to the HCV treatment and the degree of liver injury. There are three versions of the gene, a version that is corrupted by a so-called "frameshift mutation", a fully active version (IFNL4-P70) and a low-activity version (IFNL4-S70). The new results show that while there is a big difference between the effect of treatment between the IFNL4-P70 and S70 versions, both of these versions are linked to a low degree of fibrosis (liver injury). This means that the IFNL4-S70 version combines a good treatment response with a lesser degree of fibrosis.
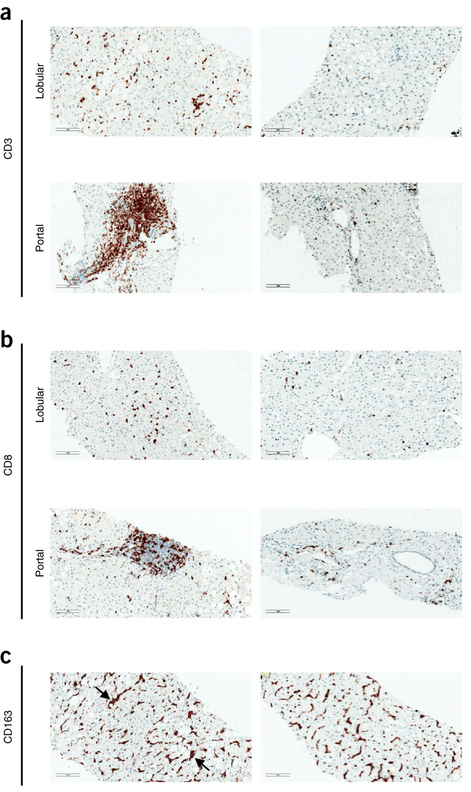
Professor Henning Grønbæk from the Medical Hepato-Gastroenterological Department V at Aarhus University Hospital also participated in the study. In a previous international collaboration with Professor Jacob George, markers for activated macrophages have been studied in patients with chronic HCV infection. Macrophages play a crucial role in inflammation and fibrosis in chronic hepatitis C infection, and measurement of CD163 in a blood sample can accurately predict the degree of inflammation and fibrosis in the liver. In the new study, blood samples were examined from 506 patients, and a correlation was found between the IFNL genotype and the amount of sCD163 and fibrosis in the liver. This confirms an interesting link between IFNL genotype, macrophage activation and risk of development of inflammation and fibrosis. At the same time, it opens up for opportunities using the sCD163 biomarker in future studies to assess the severity of the disease as well as effects of antiviral treatments.
Finally, this study opens up for an interesting scientific discussion. The Australian research team measured an increased expression of the neighbouring gene to IFNL4, IFNL3, in patients with severe liver injury, thus opening up for the possibility that the injury might be driven by the IFNL3 gene. Rune Hartmann believes that another possible interpretation could also be considered, namely that the IFNL4-S70 version of the IFNL4 gene results in effective removal of the HCV virus with simultaneous minimal liver injury. Thus, this study sets the stage for future studies to determine whether liver injury in HCV patients is driven by the IFNL3 or IFNL4 gene and what this difference in the response to treatment is, which will enable doctors to target the treatment.
The results were published in the international journal Nature Genetics.
For further information, please contact
Associate Professor Rune Hartmann
Department of Molecular Biology and Genetics, Aarhus University, Denmark
rh@mbg.au.dk - +45 2899 2578
Professor Henning Grønbæk
Department of Clinical Medicine – Medical Department V
Aarhus University Hospital, Denmark
henning.gronbaek@clin.au.dk - +45 21679281
Professor Jacob George
Storr Liver Centre
Westmead Institute for Medical Research
Westmead Hospital and University of Sydney, Australia
jacob.george@sydney.edu.au - +61 2 9845 7705
