New knowledge about cellular stress response
In a joint effort, a researcher from Aarhus University and an American researcher have uncovered how a specific network of “messenger molecules” are inhibited simultaneously in the cell during cellular stress. These findings contribute to the understanding of how the cell regulates networks of genes during cellular stress and could eventually contribute to advancing our understanding of diseases where this type of regulation is not functioning properly.

The vast majority of the cell’s messengers – called messenger RNAs (mRNAs) – are usually “translated” into protein in the cytoplasm to form important enzymes and protein factors that maintain cell life.
In a recently published study, researchers examined a class of mRNAs that contains an important element called “5'Terminal OligoPyrimidine tract” (5'TOP), which often encodes for proteins involved in the cell’s biosynthesis machinery.
Previous studies have shown that translation of all the approximately one hundred 5'TOP mRNAs is inhibited during nutrient starvation, which in turn mounts a cellular response that ensures cell survival and initially limited growth, while general “housekeeping processes” still take place.
This phenomenon has been observed, for example in tumour environments, where uncontrolled cell growth uses the cell's reserves of nutrients.
Identification of two novel regulators
Although the described selective repression (inhibition of expression) of 5'TOP mRNAs that occurs during nutrient starvation has been known for almost two decades, the protein factors that perform this function have so far remained elusive. The researchers have now identified two RNA-binding proteins called TIA-1 and TIAR, which have proved to be essential for this process.
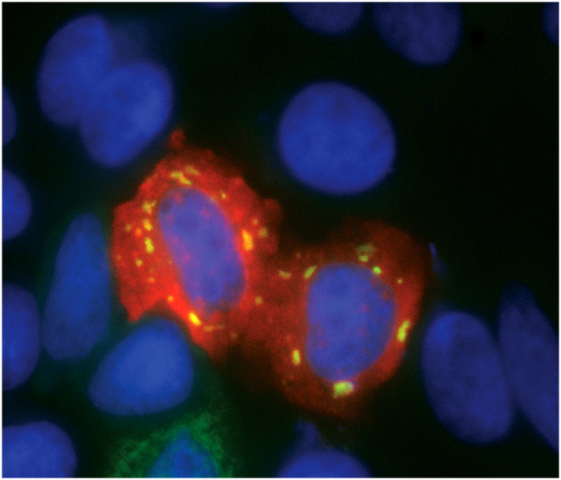
These proteins play a key role in the formation of an inhibitory complex on the 5'TOP element that inhibits translation and causes an aggregation of RNA/protein complexes in the cytoplasm. The 5'TOP mRNAs can thus accumulate in large cytoplasmic aggregates called “stress granules” (SGs) when the cell is starved of amino acids (see Figure), whereas other “ordinary” mRNAs are not affected. This correlates with the fact that 5'TOP mRNAs are no longer translated efficiently under these conditions. Furthermore, the researchers have shown that two specific cellular signalling pathways, which include the kinases mTOR and GCN2, play a crucial role in this 5'TOP mRNA regulation.
These findings further the understanding of how the cell regulates networks of genes during cellular stress and may eventually provide important insights into diseases in which such regulatory networks are mis-regulated.
The results were published this week in the renowned American journal Genes & Development and are the result of a collaboration between Associate Professor Christian Kroun Damgaard, Department of Molecular Biology and Genetics, Aarhus University, Denmark, and Associate Professor Jens Lykke-Andersen, University of California San Diego (UCSD), USA.
Link to the article "Translational coregulation of 5'TOP mRNAs by TIA-1 and TIAR"
Christian Kroun Damgaard1,2and Jens Lykke-Andersen2,3
1Department of Molecular Biology and Genetics, Aarhus University, DK-8000 Aarhus C, Denmark 2Department of Molecular, Cellular, and Developmental Biology, University of Colorado, Boulder, Colorado 80309, USA; 3Division of Biological Sciences, University of California at San Diego, La Jolla, California 92093, USA;
More information
Associate Professor Christian Kroun Damgaard
Department of Molecular Biology and Genetics, Aarhus University, Denmark
cd@mb.au.dk, +45 2970 0599
Tekst: Christian K. Damgaard and Lisbeth Heilesen
Translation: Lisbeth Heilesen
