A small protein with many applications
Researchers from the Department of Molecular Biology and Genetics and from the Department of Biomedicine at Aarhus University have collaboratively developed and described a llama-antibody that might have significant impact for future diagnostics and treatment of, e.g., kidney diseases.
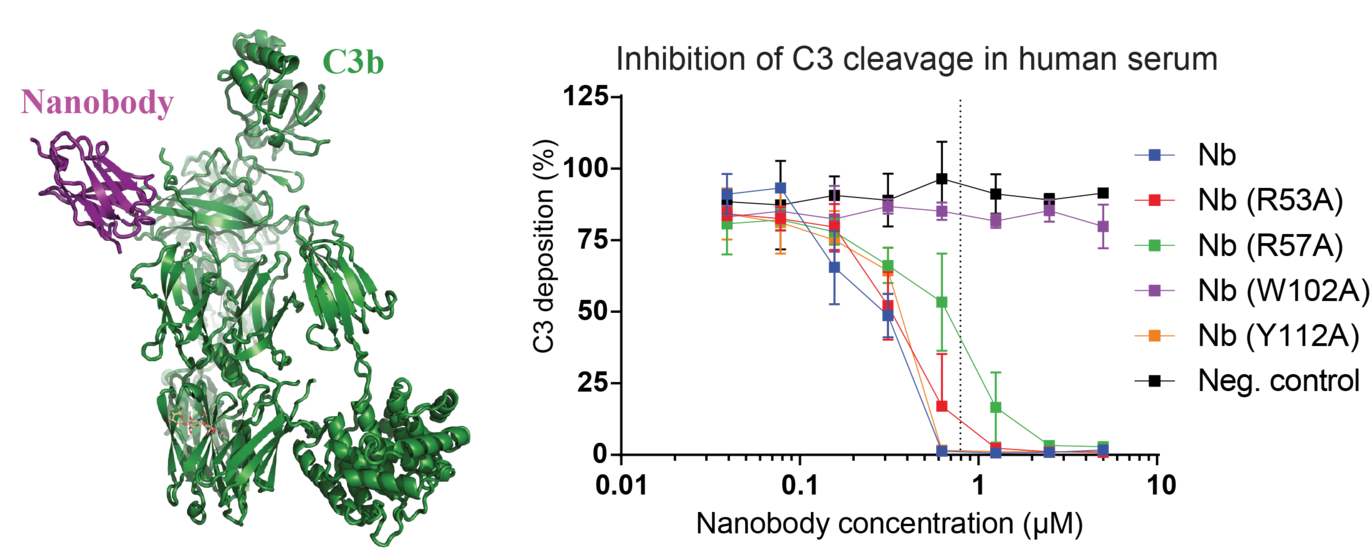
The research team has studied an important protein, called C3, from the part of the innate immune system known as the complement system. Upon recognition of pathogenic organisms or dying cells from our own body, C3 is cleaved by blood enzymes as part of a defence mechanism. These enzymes are known as C3 convertases, and the cleavage of C3 results in two fragments. The fragment called 3b binds directly to the pathogens, while the other fragment recruits and stimulates immune cells to eliminate the wrongdoer.
In a healthy person, our own cells are protected against the effects of the C3 cleavage. However, as a result of mutations in our DNA, this protection might be lost, increasing the risk of developing chronic inflammatory disorders or autoimmune diseases. For a long time, the researchers have been focussing on developing agents that prevent such undesirable complement activation. For this reason, they developed the llama-antibody to prevent cleavage of C3. Llama-antibodies are also known as nanobodies due to their small size, and compared to normal antibodies, they are easy to develop and significantly cheaper to produce.
”The technology used to develop and produce these nanobodies has been known for more than two decades” says Gregers Rom Andersen, and adds ”and therefore it is somewhat surprising that we, as far as we know, are the first in the world to develop a nanobody that inhibits the complement system.”
By exposing crystals to X-rays and correlating the results with data from electron microscopy, the researchers have described how their nanobody binds C3 in large details.
”The crystal structure of our nanobody bound to C3 supports our earlier proposed models describing how the complement convertases recognize their substrates and therefore clearly explains why our nanobody exerts its inhibitory effect on the cleavage of C3. Our results also improve the basic understanding of how this essential mechanism within the complement system works. By comparing with previously published protein structures, we can furthermore explain why our nanobody inhibits the formation of the C3 convertases, our nanobody has a ’double inhibitory’ effect so to speak,” explains Rasmus Kjeldsen Jensen.
The newly developed nanobody inhibits the cleavage of the C3 protein
In addition to the work describing the C3-nanobody complex, the research team has also performed several laboratory experiments using serum from both mice and humans. These results confirm the observations from the atomic structure and clearly reveal that their newly developed nanobody inhibits the cleavage of C3 in both human and murine serum. The latter is important, since mice are often used as disease models.
”Our experiments in serum are important as they show that our nanobody actually works under physiological conditions, where the complement system naturally exerts its function. These results indicate that our nanobody could be used in treatment of diseases caused by overactivation of the complement system. As an example, part of the kidney is not protected against complement to the same degree as other tissues, and in this case our nanobody could be a candidate to stop complement driven disease progression,” says Gregers Rom Andersen. ”However, we also found that our nanobody is able to inhibit the protective effects our own body normally wield against complement, and one should therefore be very careful and meticulously assess the possible side effects of therapeutic treatment of diseases using our nanobody,” concludes Gregers Rom Andersen.
The foundation of this work was established by the two Assistant Professors Kasper Røjkjær Andersen and Nick Stub Laursen from the Department of Molecular Biology and Genetics, who were the first in Denmark to systematically develop and produce large-scale nanobodies. However, the driving force in the project was PhD student Rasmus Kjeldsen Jensen under the supervision of Professor Gregers Rom Andersen, who in the characterization of the nanobody was assisted by the two PhD students Trine Amalie Fogh Gadeberg and Rasmus Pihl.
The results have been published recently in the reputable journal: Journal of Biological Chemistry:
Rasmus K Jensen, Rasmus Pihl, Trine A.F. Gadeberg, Jan K. Jensen, Kasper R. Andersen, Steffen Thiel, Nick S Laursen and Gregers Rom Andersen: A potent complement factor C3 specific nanobody inhibiting multiple functions in the alternative pathway of human and murine complement.
De opnåede resultater er netop blevet offentliggjort i det anerkendte tidsskrift Journal of Biological Chemistry:
Rasmus K Jensen, Rasmus Pihl, Trine A.F. Gadeberg, Jan K. Jensen, Kasper R. Andersen, Steffen Thiel, Nick S Laursen and Gregers Rom Andersen: A potent complement factor C3 specific nanobody inhibiting multiple functions in the alternative pathway of human and murine complement.
Background information
For further information, please contact
Professor Gregers Rom Andersen
Department of Molecular Biology and Genetics
Aarhus University, Denmark
gra@mbg.au.dk
+45 30256646
